 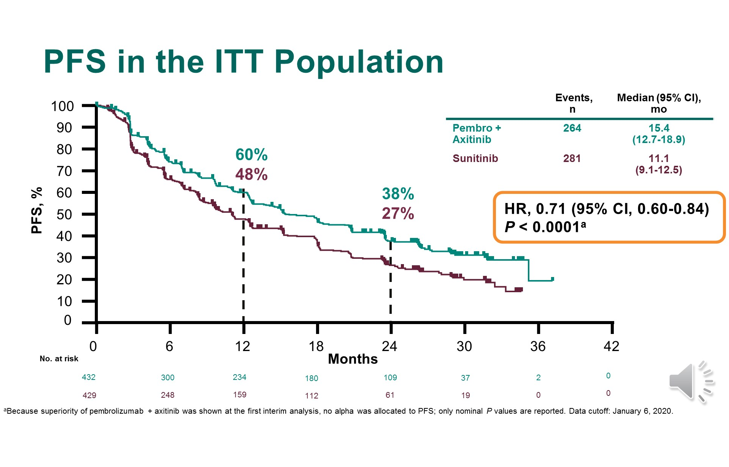
Keytruda’s sales advantage over rival immunotherapy Opdivo is based around its approval in the lucrative first-line lung cancer indication, where BMS’ drug has failed to produce convincing trial results.īut US-based Merck & Co is trying to press home this advantage by getting Keytruda (pembrolizumab) approved in a range of other cancer indications, including those where Opdivo (nivolumab) already has a foothold. Version 3.2021.Merck & Co and its cancer immunotherapy Keytruda has gained a double boost in its tussle with rival Bristol-Myers Squibb, with positive results in kidney cancer, and the promise of an FDA fast review in head and neck cancer. Phase 3 trial of lenvatinib (LEN) plus pembrolizumab (PEMBRO) or everolimus (EVE) versus sunitinib (SUN) monotherapy as a first-line treatment for patients (pts) with advanced renal cell carcinoma (RCC) (CLEAR study). FDA approves pembrolizumab plus axitinib for advanced renal cell carcinoma. Merck’s KEYTRUDA® (pembrolizumab) Demonstrated Superior Disease-Free Survival (DFS) Compared With Placebo as Adjuvant Therapy in Patients With Renal Cell Carcinoma (RCC) Following Surgery. The combination also significantly improved PFS versus sunitinib, with a median PFS of 23.9 months versus 9.2 months, respectively (HR, 0.39 P <.001).īased on the findings from the CLEAR trial, the National Comprehensive Cancer Network added pembrolizumab/lenvatinib to its clinical guidelines as a Category 1 recommendation for the frontline treatment of patients with advanced RCC. 3 The median OS was not reached in either arm. 0001).Īlso in the frontline RCC setting, it was reported earlier this year at the 2021 Genitourinary Cancers Symposium that pembrolizumab plus lenvatinib (Lenvima) reduced the risk of death by 34% versus sunitinib in the phase 3 CLEAR trial. The combination also led to significant improvement in progression-free survival (PFS), with a median PFS of 15.1 months versus 11.1 months with sunitinib (HR, 0.69 P =. 
The median OS was not reached in either group of patients. The 12-month OS rates were 90% versus 78% with the combination versus sunitinib, respectively. The data from KEYNOTE‑426 evaluated by the FDA showed that pembrolizumab/axitinib reduced the risk of death by 47% versus sunitinib in the frontline setting (HR, 0.53 P <.0001). 2 The approval was based on the phase 3 KEYNOTE‑426 trial (NCT02853331). Pembrolizumab is currently approved by the FDA for use in combination with axitinib as a frontline treatment for patients with advanced RCC. Patients were not eligible to enroll if they had prior radiotherapy for RCC, or brain or bone metastatic lesions. The parallel assignment, double-blind phase 3 KEYNOTE-564 trial (NCT03142334) included patients with RCC with a clear cell component who had undergone nephrectomy and had intermediate-high risk, high risk, or M1 no evidence of disease (M1 NED). We look forward to sharing results of KEYNOTE-564 with the medical community and regulatory authorities as soon as possible,” added Baynes. “These new data are the result of our research to evaluate the role of Keytruda in helping patients with earlier stages of disease and are the first positive results for an anti-PD-1 therapy in the adjuvant treatment of patients with renal cell carcinoma. “Since its first approval in renal cell carcinoma nearly 2 years ago, Keytruda has become an important first-line treatment option in combination with axitinib for patients with advanced renal cell carcinoma,” said Roy Baynes, MD, PhD, senior vice president and head of global clinical development, chief medical officer, Merck Research Laboratories, stated in a press release. The company plans to present the actual data from the study at a future medical conference. The trial remains ongoing to evaluate the key secondary end point of overall survival (OS).Īccording to Merck, no new safety signals emerged as compared with previously reported trials of pembrolizumab. 1Īn independent data panel determined during an interim analysis that, as compared with placebo, single-agent pembrolizumab achieved the trial’s primary end point by inducing a statistically significant and clinically meaningfully DFS improvement. Adjuvant pembrolizumab (Keytruda) extended disease-free survival (DFS) in patients with renal cell carcinoma (RCC), according to findings from the phase 3 KEYNOTE-564 trial announced today by Merck (MSD), the developer of the PD-1 inhibitor.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
